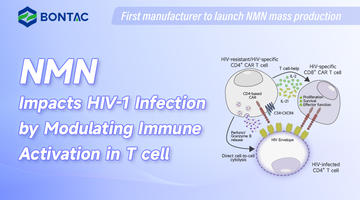
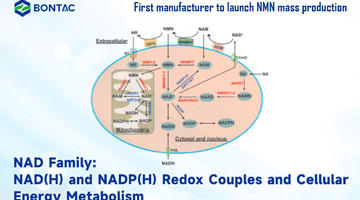
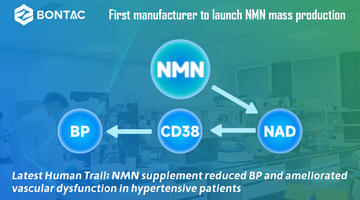
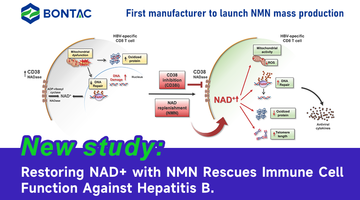
Suplemen NMN: Perisai Terhadap Fibrosis Pelbagai Organ yang Disebabkan oleh Doxorubicin

Pengenalan
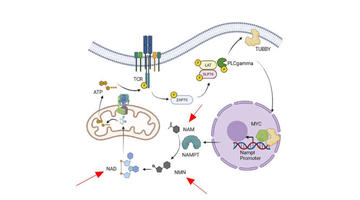
Dalam tetapan klinikal, doxorubicin (DOX) ialah ubat anti-kanser spektrum luas yang berkesan dalam kemoterapi untuk pelbagai tumor, termasuk keganasan hematologi dan tumor pepejal seperti leukemia, limfoma, kanser payudara, ovari dan paru-paru. Walau bagaimanapun, terdapat halangan yang menggerunkan untuk penggunaan DOX secara meluas, iaitu kesan sampingan toksiknya pada organ penting seperti jantung, hati dan paru-paru. Menariknya, kesan sampingan toksik DOX berkait rapat dengan penglibatan nicotinamide adenine dinucleotide (NAD+). Untuk tujuan ini, menambah nicotinamide mononucleotide (NMN), prekursor NAD+, mungkin merupakan pendekatan yang berkesan untuk mengurangkan ketoksikan DOX.Mengenai DOX
DOX, yang diasingkan daripada bakteria tanah, Streptomyces peucetius, telah ditemui pada tahun 1969 sebagai homolog daunorubicin. Walaupun keberkesanannya yang kuat, pentadbiran DOX jangka panjang boleh menyebabkan kerosakan teruk pada banyak organ dalam badan, dengan jantung menjadi sasaran utama ketoksikan doxorubicin. DOX mempunyai kardiotoksisiti yang bergantung kepada dos, termasuk disfungsi ventrikel kiri, pembentukan semula ventrikel kiri progresif dan kegagalan jantung. Yang penting, pengumpulan DOX boleh menyebabkan fibrosis yang memainkan peranan penting dalam perkembangan patologi pelbagai organ.
Kecederaan pelbagai organ yang disebabkan oleh DOX pada tikus
DOX membawa kepada ketoksikan kepada pelbagai organ, dengan kesan yang paling ketara ke atas jantung, hati dan paru-paru. Secara morfologi, penurunan ketebalan pada dinding posterior ventrikel kiri (LVPW), seni bina miokardium yang musnah, edema dalam sel hepatik dan ruang alveolar runtuh diperhatikan selepas rawatan DOX. Selain itu, berat badan yang berkurangan (~20%) dan berat organ, bersama-sama dengan kadar kematian yang tinggi (60%), dilihat dalam suntikan selepas DOX. Selain itu, NMN menghasilkan bilangan mutlak sel stem/nenek moyang hematopoietik (HSPC), sel stem hematopoietik jangka pendek (ST-HSC) dan nenek moyang multipoten (MPP).
Kesan mitigasi NMN terhadap ketoksikan yang disebabkan oleh DOX
Suplemen NMN melemahkan ketoksikan yang disebabkan oleh DOX kepada pelbagai organ dan menggalakkan kelangsungan hidup tikus dengan meningkatkan Tahap NAD+. Secara konkrit, meningkatkan tahap NAD+ melalui suplemen NMN mengurangkan disfungsi mitokondria, kerosakan DNA dan tekanan pengoksidaan, seperti yang ditunjukkan oleh potensi transmembran mitokondria (MTP) yang tinggi dan kandungan ATP serta tahap γH2AX dan ROS yang berkurangan. Selain itu, NMN mengimbangi apoptosis fibroblas MRC5, seperti yang disokong oleh penurunan peraturan p21, penanda pro-apoptosis. Sementara itu, suplemen NMN mengurangkan fibrosis jantung, hati dan paru-paru, seperti yang dibuktikan oleh pengurangan kerosakan selular dan penyusupan makrofaj, serta penurunan peraturan α-SMA, TGF-β1 dan p-smad 2/3.
Kesimpulan
Meningkatkan tahap NAD+ melalui suplemen NMN melegakan kerosakan selular, fibrosis dan penyusupan makrofaj yang disebabkan oleh DOX, dan meningkatkan kadar kelangsungan hidup tikus, membayangkan prospek aplikasinya yang berpotensi dalam mengurangkan kesan sampingan ketoksikan ubat kemoterapeutik.
Rujukan
Wen F, Xu A, Wei W, et al. Suplemen Mononukleotida Nicotinamide Mengurangkan Fibrosis Pelbagai Organ yang Disebabkan oleh Doxorubicin. Int J Mol Sci. 2024; 25(10):5303. Diterbitkan 2024 13 Mei. doi:10.3390/ijms25105303BONTAC NAD
BONTAC telah berdedikasi untuk R&D, pembuatan dan penjualan bahan mentah untuk koenzim dan produk semula jadi sejak 2012, dengan kilang milik sendiri, lebih daripada 170 paten global serta pasukan R&D yang kuat. BONTAC mempunyai pengalaman R&D yang kaya dan teknologi canggih dalam biosintesis NAD dan prekursornya (cth. NMN dan NR). Terdapat pelbagai jenis NAD yang boleh dipilih, merangkumi Gred NAD ER (penyingkiran endoksin), Gred I NAD (IVD/makanan tambahan/serbuk mentah kosmetik), Gred II NAD (API/perantaraan) dan NAD Gred IV (jika ada keperluan yang lebih tinggi pada keterlarutan), yang boleh disediakan dalam bentuk serbuk liofilisasi atau serbuk kristal. Ketulenan BONTAC NAD boleh mencapai melebihi 98%.